alb3800502
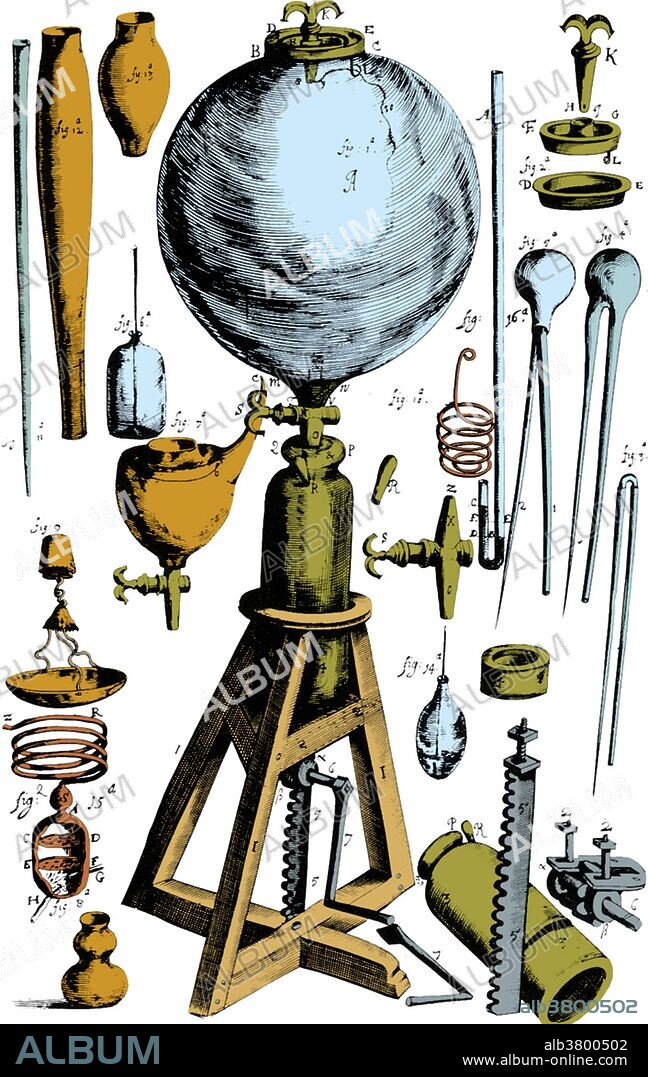
Robert Boyle, Experimental Air Pump
|
Ajouter à une autre Lightbox |
|
Ajouter à une autre Lightbox |



Avez-vous déjà un compte? S'identifier
Vous n'avez pas de compte ? S'inscrire
Acheter cette image.
Sélectionnez l'usage:

Titre:
Robert Boyle, Experimental Air Pump
Légende:
Voir la traduction automatique
Among Boyle's earliest scientific work were studies involving the air pump. At the time, Robert Hooke was Boyle's laboratory assistant. Starting with the German physicist Otto von Guericke's description of an air pump, Hooke improved on its design, reducing its size and increasing its performance while making it easier to use. Utilizing this improved air pump, Boyle devised experiments to explore the properties of air. He examined the behavior of sound, heat, light, electricity, magnetism, chemical reactions (such as a flame), and living systems (such as small animals or plants) in a vacuum. He also considered the behavior of the air itself under extension or compression. The result of this study was the relationship now known as Boyle's law, which states that the pressure and volume of a confined air (gas) are inversely related. Mathematically, this is expressed as pressure times volume equals a constant: PV = constant.
Crédit:
Album / Science Source
Autorisations:
Modèle: Non - Propriété: Non
Questions sur les droits?
Questions sur les droits?
Taille de l'image:
4200 x 6639 px | 79.8 MB
Taille d'impression:
35.6 x 56.2 cm | 14.0 x 22.1 in (300 dpi)
Mots clés:
ALCHIMIE • AUTEUR • AUTEURS • CÉLÈBRE • CELEBRITE • CHIMIE • CHIMISTE • EUROPÉEN • ILLUSTRATION • INVENTEUR • IRLANDAIS • IRLANDAISE • NATURALISTE (PHILOSOPHE) • POMPE A AIR • SCIENCE: CHIMIE
 Pinterest
Pinterest Twitter
Twitter Facebook
Facebook Copier le lien
Copier le lien Email
Email